What is zeta potential?
TOP > Products > Zeta Potential Analyzer
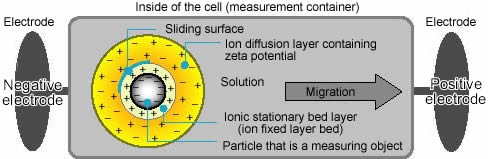
Colloidal particles dispersed in a solution are electrically charged due to their ionic characteristics and dipolar attributes.
Each particle dispersed in a solution is surrounded by oppositely charged ions called the fixed layer. Outside the fixed layer, there are varying compositions of ions of opposite polarities, forming a cloud-like area. This area is called the diffuse double layer, and the whole area is electrically neutral.
When a voltage is applied to the solution in which particles are dispersed, particles are attracted to the electrode of the opposite polarity, accompanied by the fixed layer and part of the diffuse double layer, or internal side of the "sliding surface".
 Dispersed particles |
 Aggregated particles |
Zeta potential is considered to be the electric potential of this inner area including this conceptual"sliding surface". As this electric potential approaches zero, particles tend to aggregate.
The stationary level (static layer) is computed using the following formula (which is embedded in the ZEECOM software).
The stationary level is computed using the following formula (which is embedded in the ZEECOM software).
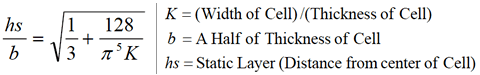
Zeta Potential (Smoluchowski’s Formula)
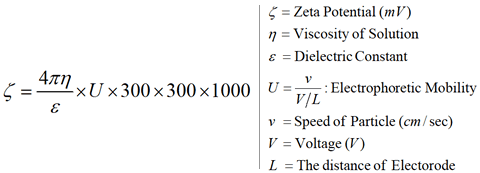
This ZEECOM unit uses a microscope to observe particle electrophoresis migration on a monitor while performing image processing to determine the particle migration speed, which is then converted into the zeta potential value.
To eliminate errors due to electroosmotic flow caused by any electrostatic charge on the measurement cell, the zeta potential is measured at the stationary level, whose position can be calculated from the width and depth of the cell. (The software performs this calculation automatically.)









